Unit . Atmosphere - Basic Structure and Atmospheric Patterns. Guided Reading Answers
The fact that the moon's surface is covered with meteorite bear on craters is obvious to the states today. Though the moon is not far from us, impact craters are few and far between on Earth. Every bit it turns out, World has received merely every bit many incoming meteorites as the moon, but the presence of the atmosphere has determined the fate of many of them. Small meteorites burn upwards in the atmosphere before always reaching Globe. Those that do hitting the surface and create an bear upon crater are lost to usa in a different way – the craters are rapidly eroded by weather generated in the atmosphere, and the bear witness is washed away. The moon, on the other manus, has no atmosphere, and thus every meteor aimed at the moon hits it, and the craters take remained essentially unchanged for 4 billion years (Figure 1).

Limerick of Earth'due south atmosphere
The early on Greeks considered "air" to be 1 of 4 uncomplicated substances; along with earth, fire, and water, air was viewed as a fundamental component of the universe. By the early 1800s, all the same, scientists such every bit John Dalton recognized that the atmosphere was in fact composed of several chemically distinct gases, which he was able to separate and determine the relative amounts of within the lower atmosphere. He was easily able to discern the major components of the temper: nitrogen, oxygen, and a small-scale amount of something incombustible, afterwards shown to be argon.
The development of the spectrometer in the 1920s allowed scientists to find gases that existed in much smaller concentrations in the temper, such as ozone and carbon dioxide. The concentrations of these gases, while modest, varied widely from place to identify. In fact, atmospheric gases are often divided up into the major, abiding components and the highly variable components, as shown in Table i and Tabular array 2.
| Nitrogen (Nii) | 78.08% |
| Oxygen (O2) | twenty.95% |
| Argon (Ar) | 0.93% |
| Neon, Helium, Krypton | 0.0001% |
Table 1: Constant Components. Proportions remain the same over fourth dimension and location.
| Carbon dioxide (COtwo) | 0.038% |
| Water vapor (H2O) | 0-4% |
| Methane (CH4) | trace |
| Sulfur dioxide (SOtwo) | trace |
| Ozone (O3) | trace |
| Nitrogen oxides (NO, NO2, N20) | trace |
Table 2: Variable Components. Amounts vary over fourth dimension and location.
Although both nitrogen and oxygen are essential to human life on the planet, they have fiddling effect on weather and other atmospheric processes. The variable components, which make up far less than 1 percent of the atmosphere, have a much greater influence on both short-term atmospheric condition and long-term climate. For example, variations in water vapor in the atmosphere are familiar to us equally relative humidity. Water vapor, CO2, CH4, NtwoO, and SO2 all have an important property: They absorb rut emitted past Earth and thus warm the temper, creating what nosotros telephone call the "greenhouse effect." Without these so-called greenhouse gases, the Earth's surface would be about xxx degrees Celsius cooler – likewise cold for life to exist every bit we know it. Though the greenhouse effect is sometimes portrayed as a bad matter, trace amounts of gases like CO2 warm our planet'due south temper enough to sustain life. Global warming, on the other paw, is a separate process that tin can be caused by increased amounts of greenhouse gases in the atmosphere.
In addition to gases, the temper as well contains particulate matter such equally dust, volcanic ash, pelting, and snow. These are, of course, highly variable and are mostly less persistent than gas concentrations, merely they tin sometimes remain in the atmosphere for relatively long periods of time. Volcanic ash from the 1991 eruption of Mt. Pinatubo in the Philippines, for case, darkened skies around the globe for over a yr.
Though the major components of the temper vary little today, they have changed dramatically over Earth'southward history, almost iv.half-dozen billion years. The early temper was hardly the life-sustaining blanket of air that it is today; most geologists believe that the main constituents then were nitrogen gas and carbon dioxide, but no costless oxygen. In fact, there is no evidence for gratuitous oxygen in the atmosphere until about 2 billion years ago, when photosynthesizing bacteria evolved and began taking in atmospheric carbon dioxide and releasing oxygen. The corporeality of oxygen in the atmosphere has risen steadily from 0 percent ii billion years ago to about 21 percent today.
Comprehension Checkpoint
Nitrogen and oxygen, which make up more than 99% of Earth's atmosphere, take a bigger influence on climate than other components of the temper.
Measuring the temper
We at present take continuous satellite monitoring of the atmosphere and Doppler radar to tell us whether or non we will experience rain anytime before long; yet, atmospheric measurements used to exist few and far between. Today, measurements such equally temperature and pressure not only aid us predict the conditions, but also assist usa expect at long-term changes in global climate (come across our Temperature module). The showtime atmospheric scientists were less concerned with weather prediction, yet, and more than interested in the composition and construction of the atmosphere.
The two about important instruments for taking measurements in Earth'southward atmosphere were adult hundreds of years ago: Galileo is credited with inventing the thermometer in 1593, and Evangelista Torricelli invented the barometer in 1643. With these two instruments, temperature and pressure could be recorded at any time and at any place. Of course, the earliest pressure level and temperature measurements were taken at Earth'south surface. It was a hundred years before the thermometer and barometer went aloft.
While many people are familiar with Ben Franklin'due south kite and key experiment that tested lightning for the presence of electricity, few realize that kites were the main vehicle for obtaining atmospheric measurements above Earth'southward surface. Throughout the 18th and xixthursday centuries, kite-mounted instruments collected pressure, temperature, and humidity readings; unfortunately, scientists could only accomplish up to an distance of almost 3 km with this technique.

Unmanned balloons were able to take measurements at higher altitudes than kites, but considering they were simply released with no passengers and no strings fastened, they had to be retrieved in order to obtain the information that had been collected. This inverse with the evolution of the radiosonde, an unmanned balloon capable of achieving high altitudes, in the early 1930s. The radiosonde included a radio transmitter amidst its many instruments, allowing information to be transmitted every bit it was being collected so that the balloons no longer needed to be retrieved. A radiosonde network was adult in the Usa in 1937, and continues to this twenty-four hour period under the auspices of the National Weather Service.
Comprehension Checkpoint
What was an advantage of the radiosonde over before information collection instruments?
Temperature in the atmosphere
Through examination of measurements collected by radiosonde and aircraft (and afterwards by rockets), scientists became enlightened that the atmosphere is not uniform. Many people had long recognized that temperature decreased with altitude – if you've ever hiked up a tall mountain, you might larn to bring a jacket to wear at the acme even when it is warm at the base – only it wasn't until the early 1900s that radiosondes revealed a layer, about 18 km in a higher place the surface, where temperature abruptly changed and began to increment with altitude. The discovery of this reversal led to partition of the atmosphere into layers based on their thermal properties.
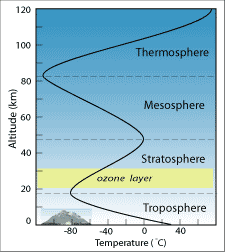
The lowermost 12 to eighteen km of the atmosphere, called the troposphere, is where all conditions occurs – clouds form and precipitation falls, air current blows, humidity varies from identify to identify, and the atmosphere interacts with the surface below. Within the troposphere, temperature decreases with altitude at a rate of about 6.5° C per kilometer. At eight,856 thou high, Mt. Everest even so reaches less than halfway through the troposphere. Bold a sea level temperature of 26° C (eighty° F), that means the temperature on the summit of Everest would be around -31° C (-24° F)! In fact, temperature at Everest's summit averages -36° C, whereas temperatures in New Delhi (in nearby India), at an elevation of 233 yard, average about 28° C (82.4° F).
At the uppermost boundary of the troposphere, air temperature reaches most -100° C and so begins to increase with distance. This layer of increasing temperature is called the stratosphere. The cause of the temperature reversal is a layer of concentrated ozone. Ozone's power to absorb incoming ultraviolet (UV) radiation from the sun had been recognized in 1881, just the beingness of the ozone layer at an altitude of 20 to 50 km was not postulated until the 1920s. By absorbing UV rays, the ozone layer both warms the air around information technology and protects the states on the surface from the harmful short-wavelength radiations that can cause pare cancer.
It is important to recognize the difference between the ozone layer in the stratosphere and ozone nowadays in trace amounts in the troposphere. Stratospheric ozone is produced when energy from the sun breaks apart O2 gas molecules into O atoms; these O atoms then bond with other O2 molecules to grade O3, ozone. This process was first described in 1930 by Sydney Chapman, a geophysicist who synthesized many of the known facts about the ozone layer. Tropospheric ozone, on the other hand, is a pollutant produced when emissions from fossil-fuel burning interact with sunlight.
Above the stratosphere, temperature begins to drib again in the next layer of the atmosphere called the mesosphere, equally seen in the previous figure. This temperature decrease results from the apace decreasing density of the air at this altitude. Finally, at the outer reaches of World's atmosphere, the intense, unfiltered radiations from the sun causes molecules like O2 and N2 to break apart into ions. The release of free energy from these reactions actually causes the temperature to rise again in the thermosphere, the outermost layer. The thermosphere extends to about 500 km above Earth's surface, still a few hundred kilometers below the altitude of nigh orbiting satellites.
Comprehension Checkpoint
All weather, including clouds, wind, and precipitation, occurs in the
Pressure in the atmosphere
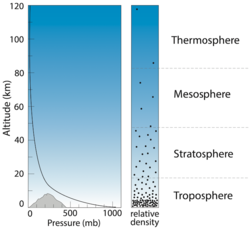
Atmospheric pressure level can be imagined equally the weight of the overlying column of air. Unlike temperature, pressure decreases exponentially with altitude. Traces of the temper can be detected every bit far as 500 km above Earth's surface, just 80 percent of the atmosphere's mass is contained within the xviii km closest to the surface. Atmospheric pressure is generally measured in millibars (mb); this unit of measurement is equivalent to one gram per centimeter squared (1 m/cm2). Other units are occasionally used, such as bars, atmospheres, or millimeters of mercury. The correspondence between these units is shown in the table below.
| bars | millibars | atmospheres | millimeters of mercury | |||
|---|---|---|---|---|---|---|
| 1.013 bar | = | 1013 mb | = | one atm | = | 760 mm Hg |
Tabular array 3: Correspondence of atmospheric measurement units.
At bounding main level, pressure ranges from about 960 to 1,050 mb, with an boilerplate of 1,013 mb. At the superlative of Mt. Everest, pressure is equally low as 300 mb. Because gas pressure is related to density, this depression pressure level means that there are approximately one-tertiary equally many gas molecules inhaled per breath on height of Mt. Everest as at body of water level – which is why climbers experience ever more severe shortness of breath the college they go, every bit less oxygen is inhaled with every breath.
Though other planets host atmospheres, the presence of free oxygen and water vapor makes our atmosphere unique as far as nosotros know. These components both encouraged and protected life on Earth equally it developed, not only by providing oxygen for respiration, but by shielding organisms from harmful UV rays and by incinerating pocket-size meteors before they hit the surface. Additionally, the limerick and structure of this unique resource are important keys to understanding circulation in the temper, biogeochemical cycling of nutrients, short-term local weather patterns, and long-term global climate changes.
Summary
Globe's atmosphere contains many components that can be measured in different ways. This module describes these different components and shows how temperature and pressure change with distance. The scientific developments that led to an understanding of these concepts are discussed.
Key Concepts
-
Earth's atmosphere is made up of a combination of gases. The major components of nitrogen, oxygen, and argon remain constant over time and infinite, while trace components like CO2 and h2o vapor vary considerably over both infinite and time.
-
The atmosphere is divided into the thermosphere, mesosphere, stratosphere, and troposphere, and the boundaries between these layers are divers by changes in temperature gradients.
-
Force per unit area decreases exponentially with altitude in the temper.
-
Our knowledge about the atmosphere has developed based on data from a variety of sources, including direct measurements from balloons and aircraft equally well equally remote measurements from satellites.
Source: https://www.visionlearning.com/en/library/Earth-Science/6/Composition-of-Earths-Atmosphere/107
0 Response to "Unit . Atmosphere - Basic Structure and Atmospheric Patterns. Guided Reading Answers"
Post a Comment